New to Wegovy®? Call NovoCare® helpline

Congratulations on taking the first step in your
Wegovy® journey.
Your pack contains a FlexTouch® pen and 4 NovoFine® Plus needles. It is to be taken on the same day of each week, at any time of day, with or without food. Each pen contains 4 doses and will last you 4 weeks.
4 doses in
each pen
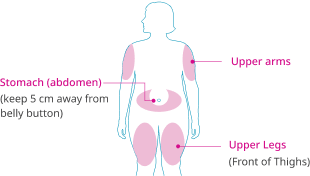
You can inject Wegovy® in 3 areas. You may inject in the same area each week, but not in the exact same spot each time.
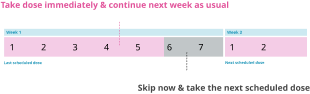
If your next scheduled dose is more than 2 days (48 hrs) away → Take the missed dose as soon as you remember.
If your next scheduled dose is less than 2 days away → Skip it and take the next dose on the scheduled day
If you have more 2 or more consecutive doses → Speak to your doctor before restarting
Keep the pen in the fridge (2 °C to 8 °C) in the original packaging to protect it from light.
Do not Freeze
After first use, you can also keep the pen at room temperature below 30 °C for up to 6 weeks
Step up safely one dose at a time. Your doctor likely prescribed Wegovy® with a
gradual
monthly increase
in dose. This helps your body adjust and keeps side effects
manageable.
Step up safely one dose at a time. Your doctor likely prescribed Wegovy® with a gradual monthly increase in dose. This helps your body adjust and keeps side effects manageable.
Some people may need more time between doses, depending on
how your
body responds your doctor will guide you.
Wegovy® doses are recommended to be taken on the same day each week.
Most common side effects are nausea and vomiting. These are mostly mild to
moderate,
resolve over time
and may not require discontinuation of Wegovy®
The important thing is to keep going, as staying on treatment is key to seeing long-term results.
The most common side effects are mild to moderate and usually go away with time, This includes nausea, diarrhoea, constipation and vomiting.
Mild to moderate side effects like nausea, vomiting, diarrhoea, or constipation are common in the beginning and they usually go away with time. In the event of severe, persistent or worsening symptoms, please reach out to your doctor.
Mild to moderate side effects like nausea, vomiting, diarrhoea, or constipation are common in the beginning and they usually go away with time.
Join the free 12-month program
Get guidance, reminders, and someone to talk to when it matters most.
Recommended for first time users.
No pressure. Just good guidance.
Your NovoCare® coach will walk you through how Wegovy® works, and answer your questions.
Regular reminders and check-ins timed to your dosing schedule—especially helpful during the first 3 months.
Learn how to manage mild side effects, improve your habits, and stay confident
From motivation to tracking progress & building lifestyle changes, your coach helps you unlock a better weight management journey
That’s why this program pairs you with a real person. Someone who checks
in,
shares answers, and helps
you stay motivated.
That’s why this program pairs you with a real
person. Someone who
checks
in,
shares answers, and helps you stay motivated.

Get all the benefits of the Novocare® Coach
Program
plus personalized
tools,
tips, and
expert
guidance all in one seamless place!
Get all the benefits of the Novocare® Coach Program plus personalized tools, tips, and expert guidance all in one seamless place!
Semaglutide injection 2.4 mg
Abbreviated prescribing information (and not full package
insert)
Generic
Name: Semaglutide Injection (0.25 mg/0.5 mg/1 mg/1.7 mg/2.4 mg), solution for injection (r-DNA Origin)
in pre-filled pen
Brand Name: Wegovy® (FlexTouch®)
Presentation: Wegovy® FlexTouch® is
available in 0.25
mg, 0.5 mg, 1.0 mg, 1.7 mg and 2.4 mg.
Indication: Weight Management: Semaglutide Injection (wegovy®) is
indicated as an adjunct to a reduced calorie diet and increased physical activity for chronic weight
management in adults with an initial body mass index (BMI) of 30 kg/m2or greater (obesity) or 27 kg/m2
or greater (overweight) in the presence of at least one weight-related comorbid condition (e.g.,
hypertension, type 2 diabetes mellitus, or dyslipidemia).
Limitations of Use: Wegovy® should not
beco-administered with other semaglutide containing products or with any other GLP- 1 receptor agonist.
The safety and effectiveness of semaglutide in combination with other products intended for weight loss,
including prescription drugs, over-the-counter drugs, and herbal preparations, have not been
established. wegovy® has not been studied in patients with a history of pancreatitis.
Established
cardiovascular disease: Semaglutide Injection (Wegovy®) is indicated to
reduce the risk of major adverse
cardiovascular events (cardiovascular death, non-fatal myocardial infarction, or non-fatal stroke) in
adults with established cardiovascular disease and either obesity or overweight.
Description: Wegovy® is
aclear and colourless solution for injection in pre-filled disposable pen. Dosing and administration:
The maintenance dose of semaglutide 2.4 mg once-weekly is reached by starting with a dose of 0.25 mg. To
reduce the likelihood of gastrointestinal symptoms, the dose should be escalated over a 16-week period
to a maintenance dose of 2.4 mg once weekly. In case of significant gastrointestinal symptoms, consid-er
delaying dose escalation until symptoms have improved. Method of administration: Subcutaneous use.
wegovy® is administered once weekly at any time of the day, with or without meals. It is to be injected
subcutaneously in the abdomen, in the thigh or in the upper arm. The injection site can be changed. It
should not be administered intravenously or intramuscularly. The day of weekly administration can be
changed, if necessary, as long as the time between two doses is at least 3 days (>72 hours). After
selecting a new dosing day, once-weekly dosing should be continued. Patients should be advised to read
the instruction for use included in the package leaflet carefully before administering wegovy®. Special
Population: No dose adjustment is required based on age. Therapeutic experience in patients ≥85 years of
age is limited. No dose adjustment is required for patients with mild or moderate renal impairment.
Experience with the use of semaglutide in patients with severe renal impairment is limited.
Semaglutide
is not recommended for use in patients with severe renal impairment (eGFR
<30 mL/min/1.73m2) including patients with end-stage renal disease. No dose adjustment is required for
patients with mild or moderate hepatic impairment. Experience with the use of semaglutide in
patients with severe hepatic impairment is limited. Semaglutide is not recommended for use in
patients with severe hepatic impairment and should be used cautiously in patients with mild or
moderate hepatic impairment. The safety and efficacy of semaglutide in children below 12 years of
age have not been estab-lished. Contraindications: Hypersensitivity to the active substance or to
any of the excipients. Special warnings and precautions: In order to improve the traceability of
biological medicinal products, the name and the batch number of the administered product should be
clearly recorded. Patients should be advised of the potential risk of dehydration in relation to
gastrointestinal side effects and take precautions to avoid fluid de-pletion. Acute pancreatitis has
been observed with the use of GLP-1 receptor agonists. Patients should be informed of the
characteristic symptoms of acutepancreatitis. If pancreatitis is suspected, semaglutide should be
discontin-ued; if confirmed, semaglutide should not be restarted. Caution should be exercised in
patients with a history of pancreatitis. In the absence of other signs and symptoms of acute
pancreatitis, elevations in pancreatic enzymes alone
are not predictive of acute pancreatitis. Semaglutide should not be used as a substitute for insulin
in patients with type 2 diabetes. Semaglutide should not be used in combination with other GLP-1
receptor agonist products. It has not been evaluated and an increased risk of adverse reactions
related to overdose is considered likely. Pa-tients treated with semaglutide in combination with a
sulfonylurea or insulin may have an increased risk of hypo-glycaemia. The risk of hypoglycaemia can
be lowered by reducing the dose of sulfonylurea or insulin when initiating treatment with a GLP-1
receptor agonist. The addition of wegovy® in patients treated with insulin
has not been evaluated. In
patients with diabetic retinopathy treated with semaglutide, an increased risk of developing
diabetic retinopathy complications has been observed. Rapid improvement in glucose control has been
associated with a temporary worsening of diabetic retinopathy, but other mechanisms cannot be
excluded. Patients with diabetic retinopathy using semaglutide should be monitored closely and
treated according to clinical guidelines. There is no experience with wegovy® in patients with type
2 diabetes with uncontrolled or potentially unstable diabetic ret-inopathy. In these patients,
treatment with wegovy® is not recommended. Use in special populations (Fertility, pregnancy and
lactation): Women of childbearing potential are recom-mended to use contraception when treated with
semaglutide. There are limited data from the use of semaglutide in pregnant women. Therefore,
semaglutide should not be used during pregnancy. If a patient wishes to become pregnant, or
pregnancy occurs, semaglutide should be discontinued. Semaglutide should be discontinued at least 2
months before a planned pregnancy due to the long half-life. Semaglutide should not be used during
breast-feeding. The effect of semaglutide on fertility in humans is unknown. Drug Interaction:
Semaglutide delays gastric emptying and could potentially influence the absorption of concomi-tantly
administered oral medicinal products.
No clinically relevant effect on the rate of gastric emptying
was ob-served with semaglutide 2.4 mg, probably due to a tolerance effect. Semaglutide should be
used with caution in patients receiving oral medicinal products that require rapid gastrointestinal
absorption. Paracetamol: Semaglutide delays the rate of gastric emptying as assessed by paracetamol
pharmacokinetics during a standardised meal test. No clinically relevant effect on paracetamol was
observed with semaglutide. No dose adjustment of paracetamol is necessary when administered with
semaglutide. Oral contraceptives: Semaglutide is not anticipated to decrease the effectiveness of
oral contraceptives as semag-lutide did not change the overall exposure of ethinylestradiol and
levonorgestrel to a clinically relevant degree, when an oral contraceptive combination medicinal
product (0.03 mg ethinylestradiol/0.15 mg levonorg-estrel) was co-administered with semaglutide.
Atorvastatin: Semaglutide did not change the overall exposure of atorvastatin following a single
dose administra-tion of atorvastatin (40 mg). Atorvastatin Cmax was decreased by 38%. This was
assessed not to be clinically relevant. Digoxin: Semaglutide did not change the overall exposure or
Cmax of digoxin following a single dose of digoxin (0.5 mg). Metformin: Semaglutide did not change
the overall exposure or Cmax of metformin following dosing of 500 mg twice daily over 3.5 days.
Warfarin & other coumarin derivatives: Semaglutide did not change overall exposure or Cmax of R- and
S-warfarin following a single dose of warfarin (25 mg), and the pharmacodynamic effects of warfarin
as measured by the inter-national normalised ratio (INR) were not affected in a clinically relevant
manner. However, cases of decreased INR have been reported during concomitant use of acenocoumarol
and semaglutide. Upon initiation of semaglutide treatment
in patients on warfarin or other coumarin
derivatives, frequent monitoring of INR is recommended. Undesirable Effects: In 4 phase 3a trials,
2,650 patients were exposed to wegovy®. The duration of the trials were 68 weeks. The most
frequently reported adverse reactions were gastrointestinal disorders including nausea, diar-rhoea,
constipation and vomiting. In general, these reactions were mild or moderate in severity and of
short duration. Other undesirable effects being delayed gastric emptying, dysgeusia, dizziness and
intestinal ob-struction. Shelf life: Before use: 36 months; After first use: 6 weeks. Store below
30°C or in a refrigerator (2°C to 8°C). Storage: Keep this medicine out of the sight and reach
ofchildren.
Store in a refrigerator (2°C to 8°C). Do not freeze and do not use wegovy® if it has
been frozen. After first use: Store below 30°C or in a refrigerator (2°C to 8°C). Keep the pen cap
on when the pen is not in use in order to protect it from light. Always remove the injection nee-dle
after each injection and store the pen without a needle attached.
Disclaimer: The abbreviated package
insert is updated from the CDSCO approved package insert (File no. BIO/IMP/24/000089 dated 25 Feb
2025). wegovy® FlexTouch®, NovoFine® and Apis bull logo is a registered trade-mark owned by Novo
Nordisk A/S and registered in Denmark. Imported by: Novo Nordisk India Private Limited,
Bangalore.
*For full prescribing information, please contact +91-8040303200 or write to us at
INAgree@novonordisk.com or reach us at Novo Nordisk India Pvt Ltd, NXT Tower-2, Floor 1&2, Embassy
Manyata Business Park,
Nagavara Village, Kasaba Hobli,
Bangalore – 560 045, India.
Note: For detailed
information on this product, please refer to full package insert*.
For the use only of registered
medical practitioner or a hospital or a laboratory The Apis bull logo is registered trademarks owned
by Novo Nordisk A/S and registered in Denmark.